Inspiring
Patented and Primed for Commercialisation.
The next generation of inhaled drug delivery spacer devices that significantly outperform dated and inefficient alternatives
- Type: Retail
Inspiring
Register your interest for this equity crowdfunding offer to get exclusive access when it goes live!
What Is Inspiring?
Inspiring is an innovative medical device company developing the next generation of inhaled drug delivery technology for treatment of respiratory diseases such as asthma. Developed by world-renowned paediatric respiratory clinician Prof. Barry Clements, and in collaboration with scientists at Australia’s Telethon Kids Research Institute and the Wal-yan Respiratory Research Centre, Inspiring has developed the first significant improvement in inhalation technology for the average user in 5 decades – the Universal Spacer System (USS).
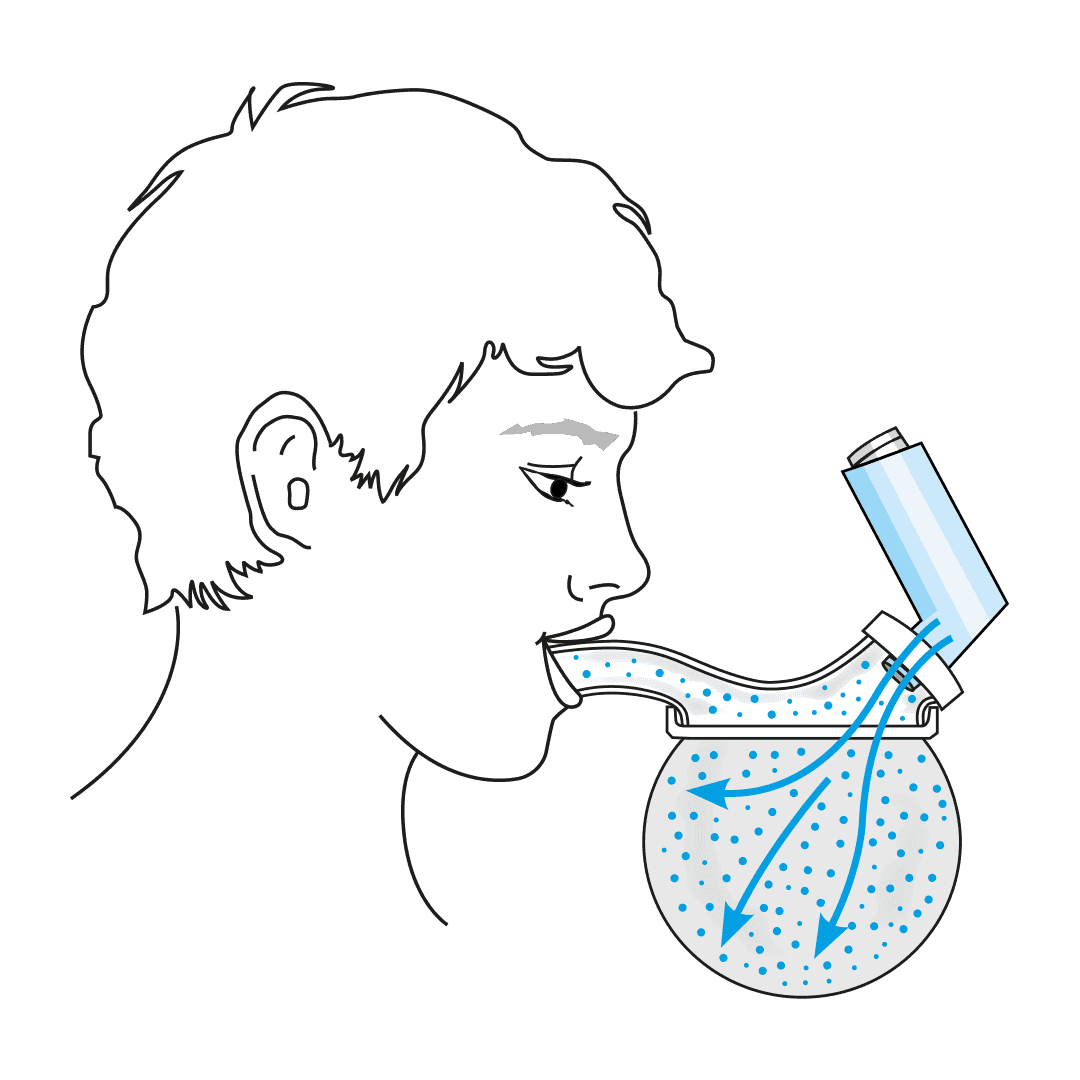
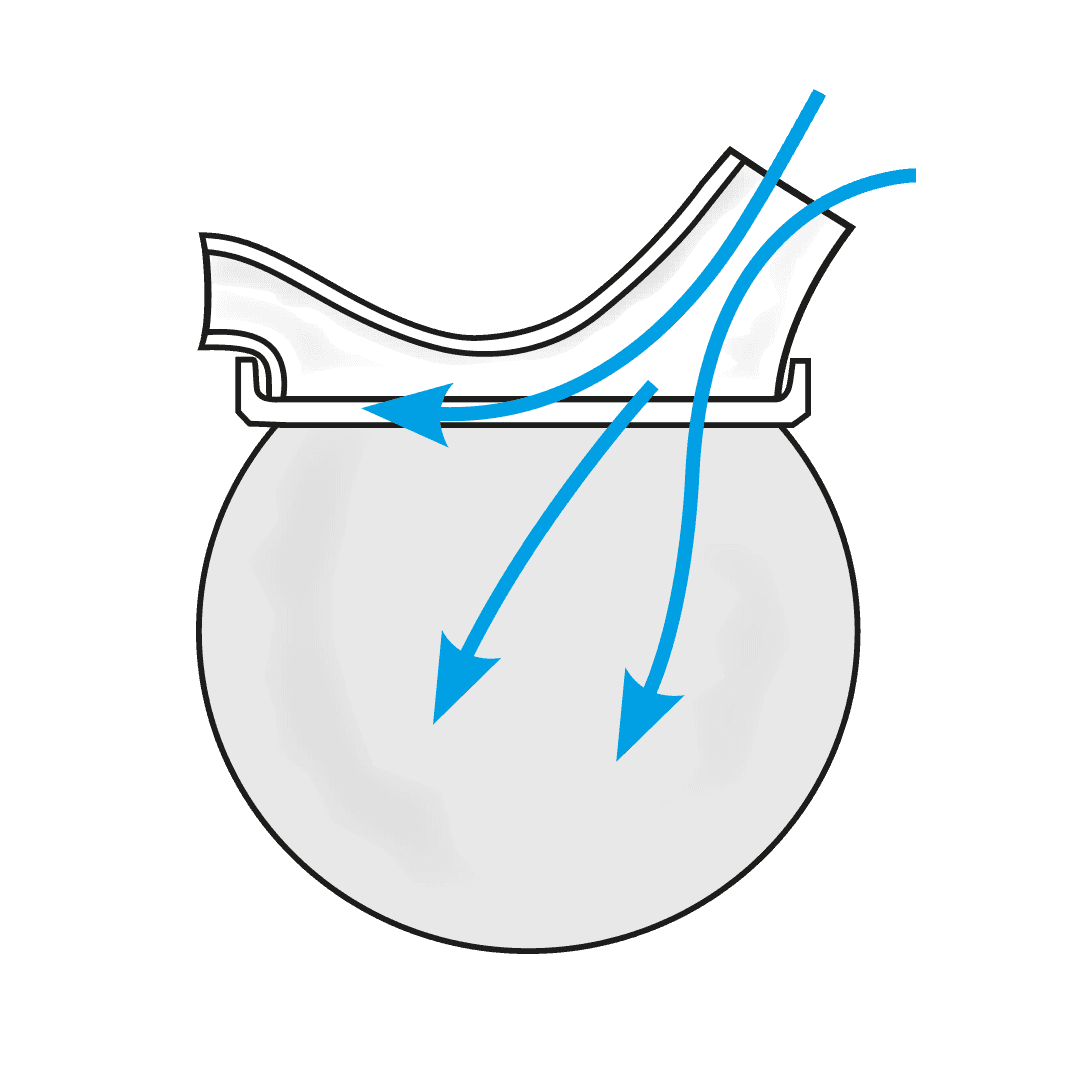
The USS is a revolutionary spacer system.
- The system resolves the issues limiting the existing spacer technology used with metered dose inhalers (MDIs or puffers) and provides a novel, collapsible, reservoir (spacer) capacity for all three inhalation drug delivery systems for the first time - MDIs, nebulisers and dry powder inhalers (DPIs).
- The USS’ simple, low-tech design will change the way inhaled drugs are delivered to the lungs, improving the lives of millions of people world-wide.
- Pre-clinical and clinical proof-of-concept testing has shown Inspiring's USS significantly outperforms current devices in all three delivery platforms in terms of efficiency (drug delivered to the lungs) and ease-of-use.
- With international patent protection for each of the MDI, DPI, and nebuliser spacers in place, the company is well-positioned to commercialise this technology.
Investment Highlights
- Next Generation Product in a Market Ready for Disruption
Inspiring’s spacer system represents the first significant improvement in inhalation technology for the average user in five decades. The $1.5B addressable MDI spacer market comprises more than 600M sufferers of asthma and chronic obstructive pulmonary disease (COPD) globally. These patients will benefit immediately from Inspiring’s USS. Inspiring’s nebuliser and DPI devices will establish new complementary markets to the $1.0B nebuliser market and $0.7B DPI market respectively.
- Strong Interest From Major International Pharmaceutical and Medical Device Manufacturers
The company has engaged in preliminary discussions with national and international medical device manufacturing companies, as well as multinational pharmaceutical companies. Each engagement has indicated a strong interest in manufacturing and commercialising Inspiring’s technology once a prototype is developed, this will likely be in the form of acquisition or licensing of Inspiring’s technology.
- Application of technology outside of asthma/COPD - including in COVID Therapies
Significant interest exists from pharmaceutical companies to deliver formulations for inhaled vaccinations, inhaled gene therapy, and other inhaled treatments with the USS. Improved efficiency and safety enables formulations that were previously not able to be delivered to become available for use only with the USS.
Inspiring is currently planning a phase one trial using the USS-Nebuliser with a novel inhaled COVID-19 treatment and is due to commence this trial in November 2020.
- Highly Experienced Management Team
An experienced board, executive and advisory team including world-renowned experts with specific respiratory experience and strong commercialisation and operational capability.
MDIs, DPIs & Nebulisers and their Existing Problems

Inhaled medications can be delivered in three ways - an MDI (puffer) producing a spray of aerosol drug, a nebuliser using a flow of air to produce a mist, and a DPI where powder containing the drug is inhaled from a pierced capsule within the device.
in over 90% of users of each device, the average percentage of dose delivered to the lung, is poor - 10% (MDI), 10-15% (DPI), to 10% (standard nebuliser) as confirmed in a number of research papers. This inefficiency impacts on patient outcomes and imposes a huge economic burden on society.
For the MDI, drug-delivery efficiency may be increased by up to 30% by attaching a spacer. However, despite spacers being universally recommended by healthcare professionals they are rigid plastic cumbersome devices which are unpopular, with only 50% of MDI users purchasing a spacer and only 10% of those purchasers using them regularly.
With the increasing incidence of respiratory diseases with population growth, especially in the elderly, there is an increasing need for improved inhalation therapy. Inspiring's USS has the potential to change the way inhaled treatment is delivered and to completely revolutionise the inhalation devices industry for the benefit of millions world-wide.
How the Universal Spacer System (USS) Addresses These Challenges
Paediatric respiratory clinician Prof. Clements, with over 40 years of respiratory clinical practice experience, knew there had to be a better way to treat his patients requiring inhaled therapy. The devastating consequences of children having asthma attacks and not being able to get enough of the drug into their lungs to relieve symptoms, are terrifying for both the patient and loved ones looking on. Since conceiving the concept, Prof. Clements has spent the past four years designing, developing, and testing a novel collapsible, closed-circuit reservoir (spacer) system that is simple, easy to use, compact, lightweight, and significantly improves ease-of-use and delivery efficiency of inhaled drugs to the lungs in all three delivery platforms. The outcome is Inspiring’s Universal Spacer System.
The USS is a patent-protected solution to inefficient and outdated treatment technology which has driven prescriber and user dis-satisfaction at an all-time high. Restoring user confidence in their inhaled medication and delivery system, will lead to increased uptake of spacers, improved patient outcomes and reduced cost, while at the same time, represents a significant commercial opportunity.
Commercialisation Plan
With three types of inhaled drug delivery systems, the Company’s first objective is to commercialise the USS MDI spacer through a licensing or acquisition of the technology. This development and commercialisation path, as well as the market conditions, are clear and well established and the projected time to market could reasonably be achieved in 2 to 3 years from the start of the program.
The COVID-19 pandemic has created significant opportunities in the inhaled delivery of new therapeutics and vaccines with demand for an efficient and safe Nebuliser delivery system. Inspiring has had a number of enquiries from pharmaceutical companies interested in using the USS nebuliser system for delivery of their inhaled product. In addition, Inspiring is about to start (November 2020) a Phase-one clinical trial with a novel inhaled COVID-19 treatment delivered using the USS Nebuliser prototype. These opportunities could lead to co-development of the USS nebuliser system in parallel with the USS MDI spacer resulting in two potential revenue streams, with a third product, the DPI variant of the device, still to be commercialised.
The pathway for each device, starting with the MDI spacer, has been carefully mapped with the development and manufacture of clinically suitable prototypes, running clinical trials, and gaining regulatory approval.
Gaining regulatory approval is a key exit milestone for medical device technologies, and achieving this milestone provides the Company with the option to either sell or license the MDI technology to a medical device company. The proceeds from the commercialisation of the MDI device will allow the Company to complete the development and commercialisation of the Nebuliser and DPI variants of the device without the need for a further capital raise in the future.
The Company having three separate products for commercialisation increases the potential for return of investment while simultaneously improving the lives of millions of people globally.
Regulatory Overview

Inspiring will follow the standard route for approval of the MDI spacer device in the first instance through:
- Therapeutic Goods Administration (TGA) in Australia;
- Food & Drug Administration (FDA) in the USA; and
- European Medicines Agency Approval in Europe.
Inspiring engaged the professional services of an international regulatory consultant to develop a report which provides a roadmap for approval with FDA and EMA with a path for the TGA to follow. The report foresees no obstacles for the standard approval along the 510K predicate device path for the MDI spacer.
FDA state approval applications are assessed within 90 days, therefore the timeframes to achieve regulatory approval for under the 510k path are not lengthy, nor onerous.
Why Equity Crowdfunding
Inspiring has decided to go down the path of equity crowdfunding to complete its capital raise owing to the consumer-based benefits provided by its technology. The company has innovated a potentially life-changing product targeted to a market of real, everyday people.
Inspiring believes that the same people who might use and benefit from its product should also have the chance to invest and share in any potential success. It is also building an engaged network of stakeholders and partners that may strategically aid the company in its growth and future development.